Accelerate Paperless Validation with ProcessX

Achieve better lifecycle management in a superior, integrated Validation Lifecycle Management (VLM) and Application Lifecycle Management (ALM) platform.
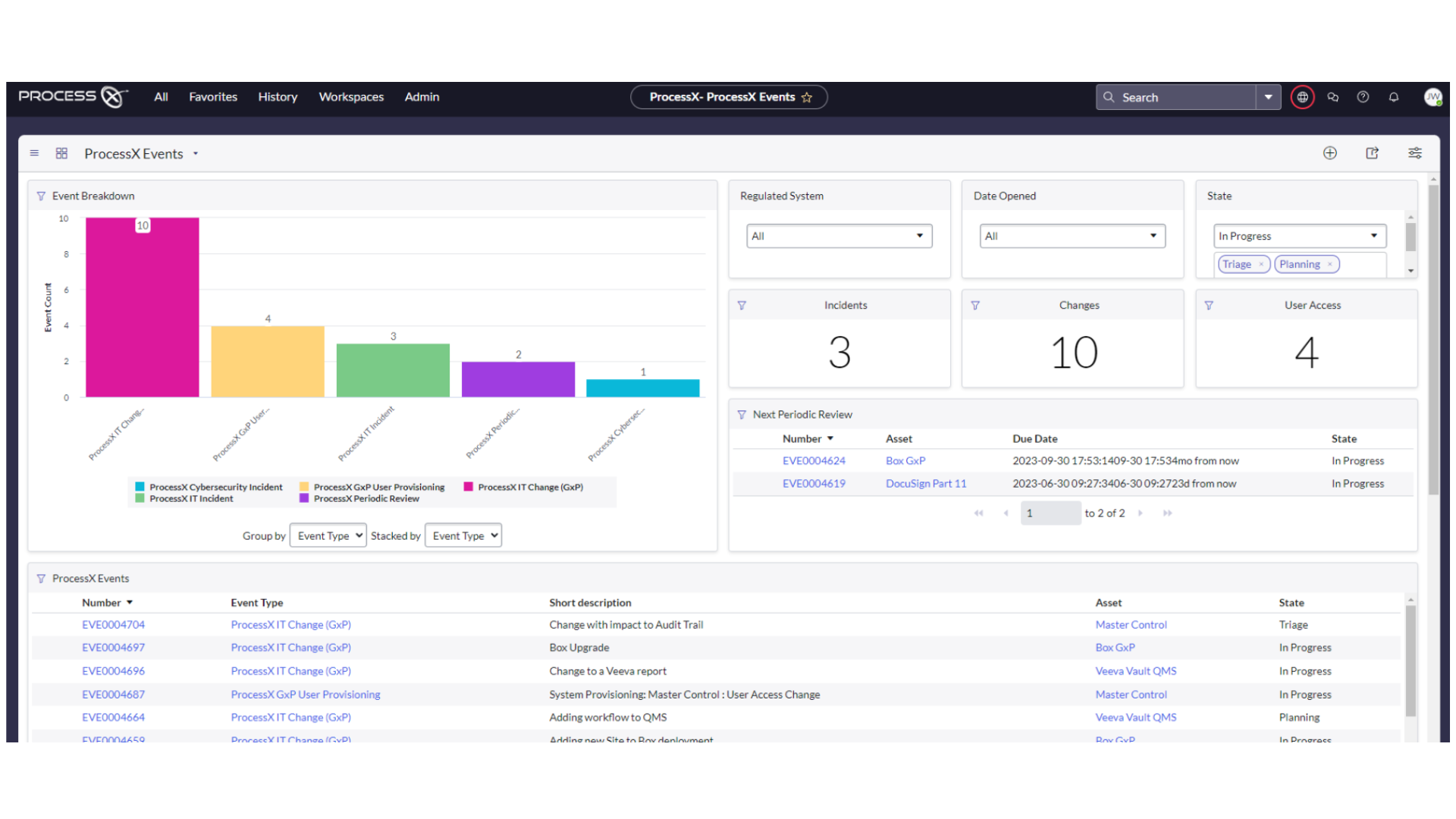
ProcessX delivers VLM, ALM, automated regression testing and release management, and GxP services to keep you continuously compliant and ahead of the pack. Paperless validation is possible today. See it for yourself.

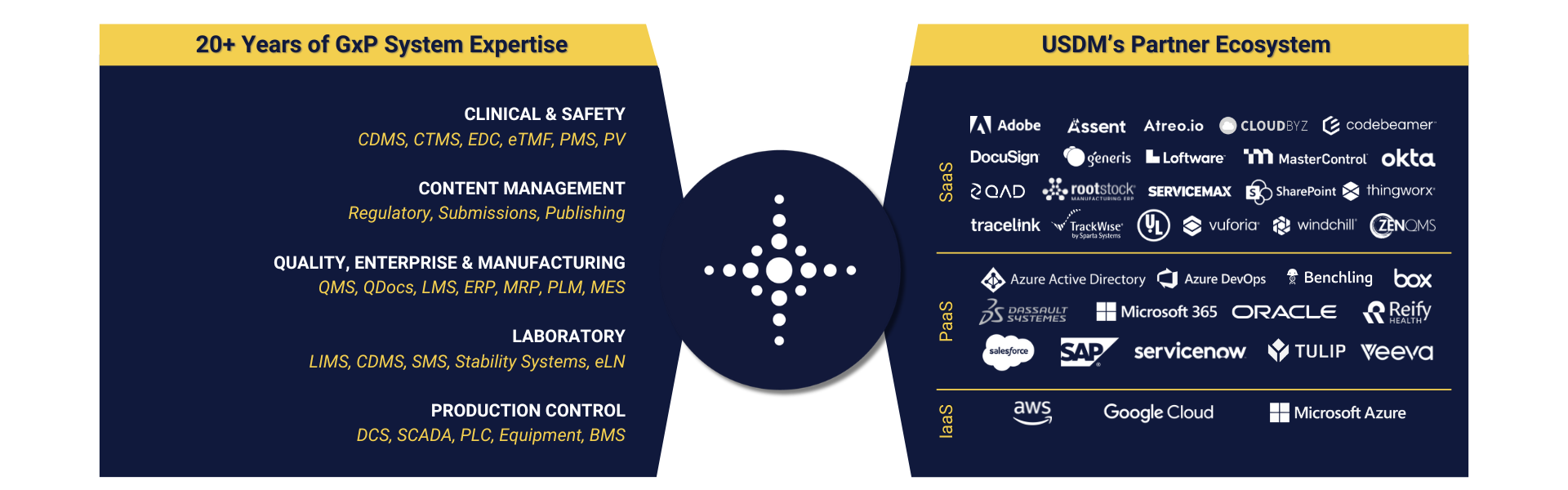
GxP and SOX Ready Capabilities
- Streamlined integration with your GxP or SOX SaaS systems
- Advanced, continuous compliance management
- Superior flexibility and customization for GxP or SOX workflows
- Enhanced user experience and citizen development
- Comprehensive GxP and SOX services and support
Business Outcomes You Can Bank On!

Reduce compliance costs by up to 80%

Harmonize Quality processes using a CSA methodology

Test Automation increases speed and efficiency by 5x

Manage all GxP, SOX and non-GxP systems using a single source of truth

Choose from dozens of audit-ready workflows
Deploy citizen development workflows from a low-code/no-code platform
AI Leaves the Pilot Phase
This clip, from USDM’s 2026 Summit, industry leaders unpack what it really means to move past experimentation and embed AI into regulated environments—without compromising confidence or audit-readiness. The conversation challenges the outdated mindset of treating AI as a one-off IT project and introduces a new model: continuous compliance, automated verification, and proactive oversight to ensure AI performance never drifts after platform updates.
If you’re responsible for quality, regulatory, IT, or digital innovation, this is a must-watch discussion on how to operationalize AI at scale—while staying always audit-ready.
Resources
Cloud Assurance as a Platform
WEBINAR
CASE STUDY
Get in touch with our team to accelerate your digital transformation today.