For most of my career, I’ve been obsessed with a simple question: How do we create massive value for life sciences organizations while still meeting the strict demands of regulation?
When you study how regulated IT operates inside most pharmaceutical and biotech companies, you discover over $1.5M per year of hidden operational waste sitting inside the way IT Quality is implemented. This isn’t due to poor performance—it’s because the systems architecture itself is flawed.
That realization is ultimately what led to the creation of ProcessX. But the story doesn’t start with technology. It starts with a problem we kept seeing over and over again across life sciences organizations.
The Hidden Friction Between IT Operations and IT Quality
Life sciences organizations operate under complex regulatory environments where every change to a GxP system must be controlled and every action must be traceable. While companies have modernized IT operations using platforms like ServiceNow, a significant problem emerges when applying GxP regulatory controls.
The reality is that ServiceNow was never designed to be a GxP-regulated system out of the box. This creates an architectural dilemma that usually forces companies down one of three inefficient paths.
ServiceNow is incredibly powerful for:
- IT Service Management (ITSM)
- Incident Management
- Change Management
- Infrastructure visibility via CMDB
But when organizations try to apply GxP regulatory controls to ServiceNow, things start to break down.
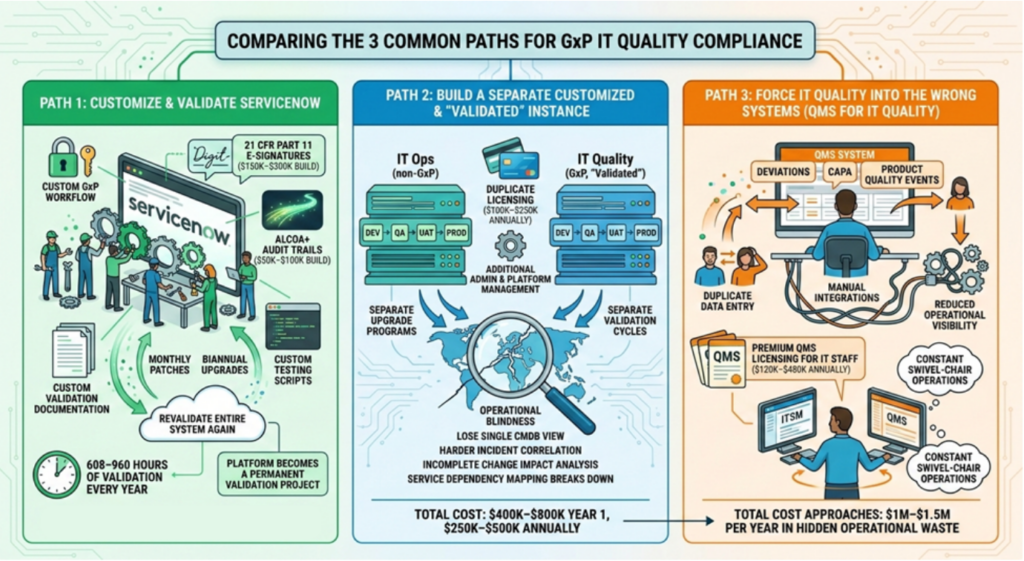
The Three Paths Companies Usually Take (And Why None Work Well)
After working with hundreds of regulated environments, I kept seeing companies choose one of three approaches. Unfortunately, each one creates significant operational friction.
Path 1: The Revalidation Trap (Customize and Validate ServiceNow)
Many organizations assume they can build regulatory capabilities themselves, but implementing real controls—like 21 CFR Part 11 electronic signatures ($150K–$300K) and ALCOA+ audit trails ($50K–$100K)—is expensive and technically demanding.
The Permanent Project: Just keeping up with monthly patches and biannual ServiceNow upgrades requires 608–960 hours of validation work every year.
The Result: The platform stops being an ITSM system and becomes a permanent validation project.
Suddenly organizations are building:
- Custom GxP workflows
- Custom audit frameworks
- Custom validation documentation
- Custom testing scripts
And every time ServiceNow upgrades? You have to revalidate the entire system again.
Path 2: Operational Blindness (Separate Validated Instances)
Some Quality teams push for a separate, validated ServiceNow environment to keep GxP data isolated. While it seems clean, the economics are significant.
Financial Burden: A second production instance requires duplicate licensing (often $100K–$250K annually) and duplicate infrastructure stacks:
- DEV → QA → UAT → PROD for each instance
- Additional environment management (upgrades, patches, account management)
- Additional administration staff
- Additional platform governance
Fragmented Visibility: When IT Operations and IT Quality live on separate instances, the organization loses the single CMDB view of their technology environment:
- Incident correlation becomes harder
- Change impact analysis becomes incomplete
- Service dependency mapping breaks down
Total Cost of Dual ServiceNow Instances
| Cost Category | Estimated Annual Impact |
|---|---|
| Duplicate licensing | $100K–$250K |
| Additional environment stack | $50K–$150K operational overhead |
| Upgrade validation effort | 4–8 weeks per major release |
| Admin & platform management | 0.5–1 FTE |
| Total Year 1 Cost | $400K–$800K |
| Total Annually Thereafter | $250K–$500K |
And yet the organization still hasn’t solved the core problem: embedding GxP controls into operational IT workflows.
Path 3: The Swivel-Chair Tax (Forcing IT Quality into QMS)
The most common approach is pushing IT Quality processes into QMS systems designed for product quality (deviations, CAPAs, batch records)—not IT operations.
But IT change control, incident management, and infrastructure risk management are IT operational processes, not product quality processes. When those processes live inside a QMS, the economics break down.
The Hidden Costs of QMS for IT Quality
| Cost Driver | Estimated Annual Impact |
|---|---|
| QMS licenses for IT staff | $3K–$8K per user annually |
| 40–60 IT users interacting with GxP systems | $120K–$480K annually |
| Duplicate data entry & manual integrations | Significant labor overhead |
| Slower change cycles & duplicate CAB meetings | 2–4 weeks added per change |
Organizations end up paying hundreds of thousands per year in QMS licensing just to run IT workflows.
The Operational Friction: Because ITSM and QMS platforms are separate, teams constantly move information between systems:
- Incidents affecting GxP systems must be manually escalated to Quality
- Change approvals require duplicate workflow steps
- System risk classification often exists outside the CMDB
The result is constant swivel-chair operations between systems.
The $1.5M Disconnect: Why IT Quality Architecture Matters
When you combine:
- QMS licensing for IT workflows
- Duplicate process steps
- Manual reconciliation during audits
- Slower change cycles
The total cost often approaches $1M–$1.5M per year in hidden operational waste. Not because teams are doing anything wrong—but because the architecture forces IT processes into systems that weren’t designed to run them.
The Insight That Led to ProcessX
The problem wasn’t ServiceNow—it’s an incredible platform. The challenge was building regulatory controls on top of it in a way that preserved the operational model rather than destroying it.
That meant solving several hard problems:
- True Part 11 electronic signatures
- ALCOA+ compliant audit trails
- Validated GxP change control
- System risk classification integrated with CMDB
- Regulatory data segregation
- Upgrade validation automation
And it had to be done in a way that did not require a second ServiceNow instance, massive customization, or continuous revalidation.
That challenge ultimately led to the creation of ProcessX—a regulatory framework designed to sit inside ServiceNow. It provides ALCOA+ compliant audit trails and 21 CFR Part 11 signatures by design, transforming ServiceNow into a GxP-ready environment without requiring a second instance or massive manual customization.
Four Approaches Compared: Which Architecture Wins?
| Dimension | Path 1: Customize ServiceNow | Path 2: Dual Instances | Path 3: QMS for IT | ProcessX |
|---|---|---|---|---|
| Architecture | Single, heavily customized | Two separate instances | Siloed ITSM + QMS | Single native instance + GxP framework |
| Single CMDB View | Yes, but modified | No – fragmented | No – siloed data | Yes – shared with GxP attributes |
| Part 11 / ALCOA+ | Custom built | Custom configured | In QMS (wrong system) | Native by design |
| Year 1 Cost | $200K–$400K+ | $400K–$800K | QMS licenses exist | Eliminates other costs |
| Annual Recurring | Massive revalidation | $250K–$500K | $1M–$1.5M hidden waste | $1.5M+ savings |
| Validation Effort | 608–960 hrs/year | 4–8 weeks/release | Manual swivel-chair | Automated, vendor-provided |
| Key Drawback | Permanent validation project | Operational blindness | Hidden costs, friction | — |
Why This Matters More Than Ever in 2026
The pressure on life sciences organizations is only increasing:
- Cybersecurity threats are growing. Regulatory scrutiny of vendor security is intensifying.
- Regulatory expectations are rising. FDA and EMA expect continuous compliance, not point-in-time validation.
- Speed-to-patient demands are higher than ever. Slower change cycles directly impact time-to-market.
At the same time, the software landscape has fundamentally changed. Most regulated systems today are SaaS platforms—they release updates constantly (monthly patches, quarterly releases, major upgrades every year).
That’s fantastic for innovation. But it creates a massive operational burden for regulated companies. Because every release triggers the same question: “Do we need to validate this again?”
In most organizations, the answer leads to a painfully manual process: impact assessments, test script execution, traceability matrices, validation reports. Over and over again.
What many companies underestimate is the sheer scale of this burden. Native ServiceNow validation alone can require hundreds of hours of annual validation work just to keep up with platform upgrades and patches. Multiply that across dozens of SaaS platforms, and the cost becomes staggering.
That’s not value creation. That’s compliance friction.
Coming Next: Cloud Assurance and the End of SaaS Validation Chaos
When we started building ProcessX, it quickly became clear that solving the GxP process problem was only part of the challenge. The bigger question:
How do you keep validated SaaS systems compliant when they update every month?
In Part 2, we’ll dive into Cloud Assurance—a hyper-efficient model designed for regulated SaaS environments. Instead of every company repeating the same manual work, Cloud Assurance centralizes the effort by providing:
- Vendor release impact assessments
- Automated validation evidence
- Pre-executed testing frameworks
- Inspection-ready validation packages
- Annual vendor audits
- Real-time vendor cyber monitoring
By deploying Cloud Assurance via ProcessX, organizations can shift from hundreds of hours of validation effort to simply reviewing vendor-provided results.
Which means teams can finally focus on what matters most: delivering innovation, improving operations, and bringing therapies to patients faster.
Frequently Asked Questions
What is the IT Quality Information Vacuum?
The IT Quality Information Vacuum is the $1.5M hidden cost that occurs when IT Quality processes are forced into systems (like QMS) that weren’t designed for IT operations. This creates duplicate data entry, manual reconciliation, slower change cycles, and premium licensing costs.
Why can’t ServiceNow be used for GxP compliance out of the box?
ServiceNow lacks native 21 CFR Part 11 electronic signatures, ALCOA+ compliant audit trails, and GxP-specific workflow controls. Customizing ServiceNow for these capabilities costs $200K–$400K+ initially, plus 608–960 hours of annual revalidation effort with each upgrade.
What is ProcessX and how does it solve this problem?
ProcessX is a regulatory framework that sits inside ServiceNow, providing ALCOA+ compliant audit trails and Part 11 electronic signatures by design. It transforms ServiceNow into a GxP-ready environment without requiring a second instance, massive customization, or continuous revalidation.
How much can organizations save by switching to ProcessX?
Organizations typically eliminate $1M–$1.5M in annual hidden operational waste, including QMS licensing for IT staff ($120K–$480K), duplicate process steps, manual reconciliation, and validation overhead.
What is the difference between IT Quality and Product Quality?
Product Quality (deviations, CAPAs, batch records) belongs in QMS systems like Veeva Vault or MasterControl. IT Quality (GxP change control, validation, incident management, system risk) belongs alongside IT Operations in ServiceNow—where the CMDB, change history, and technical context already exist.
Which Path Is Your Organization Walking?
If you’re working in regulated IT, quality, or digital transformation in life sciences, I’d love to hear your experiences:
- The Revalidation Trap — Customized & Validated ServiceNow (Path 1)
- The Data Silo — Duplicate Validated ServiceNow Instances (Path 2)
- The Swivel-Chair Tax — IT Quality Forced into QMS (Path 3)
Share your experience in the comments. And stay tuned for Part 2, where we’ll explore how to manage validation when your systems are constantly updating in the cloud.
Explore the full ProcessX series:
- Part 1: The $1.5M IT Quality Problem — This article
- Part 2: Cloud Assurance
- Part 3: Agentic AI in Regulated Environments
Related Resources
About the Author
Vega Finucan is a Co-Founder at USDM Life Sciences, where she focuses on building AI-enabled workflow solutions for regulated life sciences environments. ProcessX was born from patterns observed across hundreds of pharmaceutical and biotech organizations—and from a persistent belief that compliance shouldn’t be a tax on innovation.