Key Takeaways
Pre-commercial or recently commercial life sciences companies benefit significantly from implementing a pre-configured and pre-validated ITSM platform. These systems deliver faster time to readiness, reduced compliance risks, and scalable infrastructure to support GxP and non GxP workflows. As the business evolves, a unified ITSM approach empowers teams to stay audit-ready, drive operational excellence, and remain focused on patient impact.
Optimize Now, Scale Later
Whether you’re evaluating GxP platforms, digitizing GxP processes, or simply trying to streamline support for non GxP systems, now is the time to build an ITSM foundation that supports your growth. With platforms like ProcessX, life sciences organizations can harmonize IT operations, reduce regulatory friction, and enable a future-ready digital strategy.
Explore how ProcessX delivers compliant, scalable ITSM for life sciences.
The Problem: Navigating Complex IT Challenges in Life Sciences
For life sciences companies, IT isn’t just an operational function—it’s a strategic enabler of compliance, digital quality, and scalable growth. As organizations evolve, IT environments often become fragmented, requiring teams to manage a complex mix of GxP IT systems and non GxP platforms.
This creates a growing need to differentiate between ITSM for GxP and non GxP ITSM—each with its own set of risks, regulatory requirements, and business drivers.
The challenge? Maintaining audit-readiness, ensuring data integrity, and streamlining processes across both GxP and non GxP projects, without sacrificing speed or innovation.
Without a unified approach to IT service management, even minor disruptions, undocumented changes, or misaligned processes can compromise compliance. For emerging or pre-commercial companies, implementing a pre-validated, scalable GxP platform early helps avoid future rework and accelerates readiness for inspections, growth, and go-to-market success.
Discerning ITSM in GxP vs Non GxP Systems
While ITSM is foundational across industries, its implementation in life sciences must account for the distinction between GxP and non GxP systems.
GxP systems—such as those supporting quality, manufacturing, or clinical processes—must follow strict regulatory guidelines. ITSM in this context must incorporate GxP-aligned processes including incident tracking, change control, audit trails, and continuous compliance monitoring. A GxP ITSM platform must also support traceability, validation, and data integrity to align with FDA, EMA, and other global standards.
On the other hand, non GxP ITSM may not be directly regulated, but it still plays a critical role. Why? Because non GxP systems often integrate with GxP environments or indirectly support regulated processes. Without proper ITSM in place, gaps in these systems can introduce compliance risks.
Modern ITSM for GxP and non GxP projects—like those built into ProcessX—enable organizations to apply consistent governance, automate workflows, and create a single source of truth across their digital ecosystem. This not only simplifies audits but enhances operational efficiency, user adoption, and platform scalability.
For example, GxP process automation and GxP test automation can now be extended to traditionally non-regulated areas to improve visibility, reduce manual errors, and minimize deviations—all within an ITSM framework designed for life sciences.
Introducing ProcessX: A Unified ITSM Platform Built on the Robust Foundation of ServiceNow
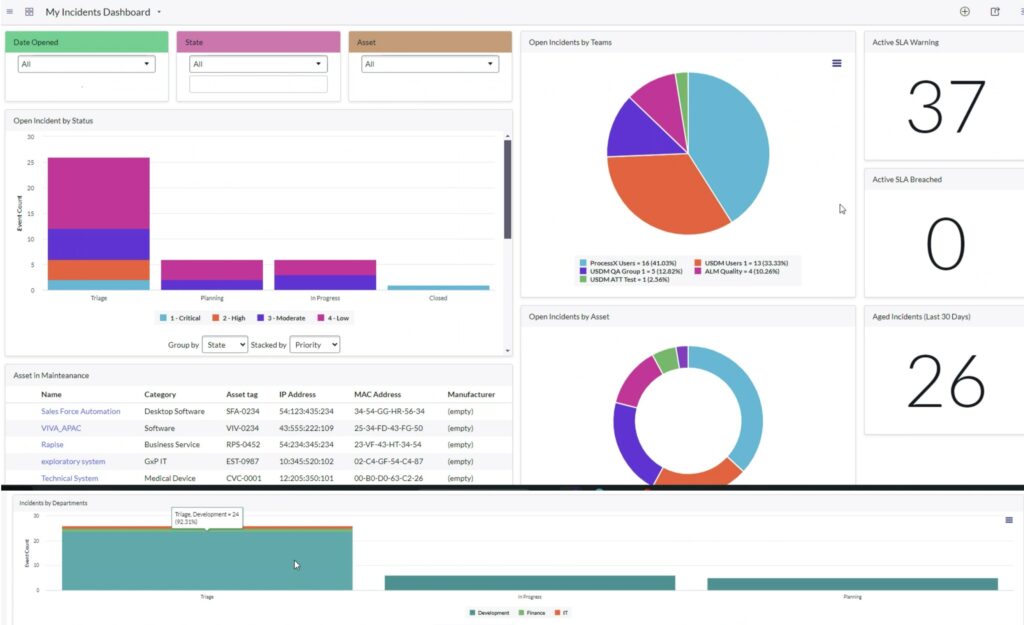
ProcessX revolutionizes IT service management by providing an all-encompassing solution that supports both non-GxP and GxP system incidents, user access requests, changes, and more. Designed on the robust platform of ServiceNow, ProcessX ensures that workflows are not only validated but remain in a state of continuous validation with each update. This integration offers IT leaders a seamless, compliant, and efficient tool to oversee their entire system landscape.
Comprehensive Features and Benefits of ProcessX
- Holistic System Management: By consolidating ticketing and change management into a single platform, ProcessX enables comprehensive oversight, reducing redundancies and improving response times.
- Pre-validated GxP Workflows: ProcessX alleviates the burden of maintaining compliance by providing pre-validated workflows that adapt to regulatory changes, ensuring your systems are always audit-ready.
- Enhanced Operational Visibility: Real-time tracking of Service Level Agreements and system statuses allows IT leaders to make informed, proactive decisions, thereby enhancing the overall effectiveness of the IT organization.
ProcessX’s Role in Streamlining Validation Activities
For GxP-regulated systems, ProcessX offers robust support through:
- Structured Validation Planning and Risk Management: Adheres to industry standards such as Computer Software Assurance and GAMP, facilitating thorough and compliant validation processes.
- Efficient Testing Capabilities: Supports both automated and manual testing to ensure comprehensive coverage and compliance.
- On-demand Traceability and Reporting: Generates real-time traceability matrices and summary reports, simplifying documentation and audit processes.
Simplifying Non-GxP IT Management
ProcessX extends its capabilities to non-GxP systems, providing streamlined workflows that handle common IT management tasks. This simplification helps maintain and manage Service Level Agreements (SLAs) effectively, ensuring consistent service quality across all IT services.
Enhancing the IT Ecosystem with Seamless Integration
ProcessX integrates flawlessly with various components of an IT ecosystem, including:
- Test Automation and Requirements Management software
- Quality Management Systems
- Content Management Systems
- Learning Management Systems
- Enterprise Resource Planning (ERP) Systems
- Customer Relationship Management (CRM) Systems
This integration is pivotal for fostering an efficient, interconnected digital ecosystem, particularly critical in environments where compliance with GxP is mandatory.
Addressing the Limitations of Traditional GxP Solutions
Traditional GxP solutions often rely on outdated paper processes or require complex, costly customizations, leading to visibility gaps and heightened compliance risks. ProcessX effectively bridges these gaps with compliant workflows, automated validation libraries, and pre-configured features that incorporate industry best practices, dramatically reducing both compliance risks and operational costs.
Invitation to Transform Your IT Operations
Are you looking to streamline your IT service management, ensure enduring compliance, and enhance operational efficiency? Reach out to the ProcessX team today to discover how our comprehensive ITSM solution can revolutionize your processes. Visit our website to download an in-depth guide on implementing ProcessX in your life sciences organization, or contact us directly to start your journey towards a simplified, compliant IT ecosystem.
Unburden your teams of disparate systems to manage your IT systems and ongoing compliance maintenance. Contact the ProcessX team today to get started today.